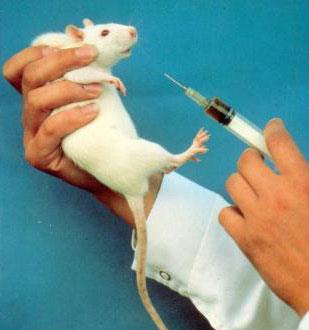
REACH
We have until 2018 to save up to 54 million animals
from being poisoned and killed
For decades compassionate, responsible consumers along with concerned organisations have been calling for an end to animal testing for cosmetics and when the EU Cosmetics Directive ban on testing cosmetics ingredients on animals came into force on 11th of March, 2013, it was a day for celebrations.
As of that date, no more new products containing ingredients tested on animals will be allowed on store shelves, regardless of where in the world the tests derived. EU’s commissioner for health and consumer policy, Tonio Borg, said in a statement that Europe wants to set an example. This ban “gives an important signal on the value that Europe attaches to animal welfare.”
The EU first got involved in anti-animal testing in 2004, when it banned testing of finished cosmetic products. Five years later it moved to ban animal-tested ingredients in all products. But after heavy lobbying from major cosmetics makers, the EU extended the deadline for some tests, mainly for allergy and cancer for which there were no substitutes.
Unfortunately, the joy over the new ban on animal testings for cosmetics, was short-lived! In fact, we can now even await a massive increase in animal experiments under another EU regulation, REACH, that deals with the Registration, Evaluation, Authorisation and Restriction of Chemical substances. This law entered into force on 1 June 2007 and it mandates that any ingredients not previously tested on animals have to undergo animal tests, totally contradicting the ban under the Cosmetics Directive and effectively spelling the end of cruelty-free cosmetics, says LUSH.
In fact, REACH legislation contains a concession that the Cosmetics Directive, which bans animal tests for cosmetics, takes precedence over REACH for cosmetic ingredients, but some organisations and cruelty-free companies such as LUSH, are concerned that a number of ingredients used in cosmetics could be tested on animals despite the ban.
There are in addition currently unresolved issues about particular types of testing that could be permitted even for cosmetics (e.g. environmental testing or worker safety), on which the ECEAE is seeking clarification.
As of that date, no more new products containing ingredients tested on animals will be allowed on store shelves, regardless of where in the world the tests derived. EU’s commissioner for health and consumer policy, Tonio Borg, said in a statement that Europe wants to set an example. This ban “gives an important signal on the value that Europe attaches to animal welfare.”
The EU first got involved in anti-animal testing in 2004, when it banned testing of finished cosmetic products. Five years later it moved to ban animal-tested ingredients in all products. But after heavy lobbying from major cosmetics makers, the EU extended the deadline for some tests, mainly for allergy and cancer for which there were no substitutes.
Unfortunately, the joy over the new ban on animal testings for cosmetics, was short-lived! In fact, we can now even await a massive increase in animal experiments under another EU regulation, REACH, that deals with the Registration, Evaluation, Authorisation and Restriction of Chemical substances. This law entered into force on 1 June 2007 and it mandates that any ingredients not previously tested on animals have to undergo animal tests, totally contradicting the ban under the Cosmetics Directive and effectively spelling the end of cruelty-free cosmetics, says LUSH.
In fact, REACH legislation contains a concession that the Cosmetics Directive, which bans animal tests for cosmetics, takes precedence over REACH for cosmetic ingredients, but some organisations and cruelty-free companies such as LUSH, are concerned that a number of ingredients used in cosmetics could be tested on animals despite the ban.
There are in addition currently unresolved issues about particular types of testing that could be permitted even for cosmetics (e.g. environmental testing or worker safety), on which the ECEAE is seeking clarification.

In this animal research testing picture, rabbits are testing the ingredients used in household products. It is an animal testing fact that there are humane alternative research methods. Help stop and ban animals being used to test household products. For your ethical, compassionate shopping guide, visit www.leapingbunny.org.
© Brian Gunn /IAAPEA
In the next video, Mark Constantine, one of the Lush founders and expert in cosmetics, talks about the history of animal testing and the implications for the cosmetic industry as the new REACH legislation lands. This is such an important issue, both for us as a vegan-vegetarian company and the entire industry; here, Mark discusses what this will mean for some cosmetic ingredients under REACH. Visit www.lush.co.uk for more information on our stance against animal testing.
What is REACH?
REACH is a new European Community Regulation on chemicals and their safe use (EC 1907/2006). Its title stands for the Registration, Evaluation, Authorisation and Restriction of Chemical substances. The aim of REACH is to improve the protection of human health and the environment through the better and earlier identification of the risky nature of chemical substances.
What has REACH got to do with animal testing?
REACH seeks to assess the safety of chemicals for human health and the environment, so tests need to be carried out to see what impact they have. The experiments used to assess the safety of chemicals are called toxicity tests, which traditionally involve the poisoning of guinea pigs, rabbits, birds, fish, rats and mice, although many non-animal alternatives now exist.
How many animals will be used in chemical safety tests?
Between 13 and 54 million animals could be used in the EU under REACH to test the safety of an estimated 30,000 or more chemicals between 2009 and 2018.
The number of animals used in cruel poisoning tests could have been much higher than the 13 million currently estimated by the European Commission - Their original proposals would have seen the poisoning of 45 million animals under REACH.
But after five years of some of the most intense lobbying and campaigning the ECEAE has ever undertaken, we were successful in securing life-saving amendments to the legislation. As a result, millions of animals were saved from suffering and death for REACH.
Unfortunately, more recent estimates of the numbers of chemicals needing to be tested have meant that the number of animals could be much higher than previously thought, despite these important amendments.
What kinds of animal experiments are carried out to test chemicals?
Toxicity tests involve force-feeding animals high doses of chemicals – many become seriously ill. The animals are always killed as part of the test if they do not die as a result of it. Tests may involve two-generation studies where pregnant animals are poisoned so the effects of the chemical can be seen in the babies.
The first proposed test under REACH was for an industrial lubricant, and involved force-feeding pregnant rats high doses of chemicals and killing them just before they are due to give birth.
If we don't test on animals how can we ensure chemicals are safe?
The ECEAE believes animals do not need to be used for chemicals testing, and published a report entitled ‘The Way Forward: A Non-Animal Testing Strategy for Chemicals’, which demonstrated how chemicals could be safely tested without having to use animals.
Ways to avoid animal tests include using humane alternatives, looking at human safety data and avoiding duplication of animal testing by sharing data on chemical tests that have already been carried out.
How can we stop animal testing from taking place now that the legislation has been passed?
REACH legislation states that some of the test proposals that are submitted to the European Chemicals Agency (ECHA) must be open to public scrutiny for 45 days. This gives ECEAE scientists a window of opportunity to provide information from other sources which might prevent the test happening, and save animals from awful suffering and death. We've already been able to suggest alternatives to the first animal testing proposals - we will have commented on all 21 proposals by the end of 2010 - and will be scrutinising further proposals as they are released.
Are the chemical companies using alternatives to animal tests?
Thanks to five years of intensive ECEAE lobbying, the use of alternatives has been placed centrally in the legislation. Article 1 specifically states that one of the aims of REACH is the ‘promotion of alternative methods of assessment of hazards of substances.’
This means that the chemical companies must use alternatives to animal tests where they are available, and puts an impetus on the European Union to develop and validate new alternatives. The ECEAE will be paying close attention to ensure that alternatives to cruel animal poisoning tests are used whenever possible. A huge shift in culture is still needed.
Cosmetics have chemicals in them, so how does REACH effect the cosmetics testing ban?
REACH legislation contains a concession that the Cosmetics Directive, which bans animal tests for cosmetics, takes precedence over REACH for cosmetic ingredients.
On this basis, we argue that this means that if a substance was animal-tested for any reason under REACH from March 2009 (when the Cosmetics Directive came into force), it could not subsequently be used in a cosmetic. However, we have a real battle on our hands to make sure that the legislation is applied properly.
There are in addition currently unresolved issues about particular types of testing that could be permitted even for cosmetics (e.g. environmental testing or worker safety), on which the ECEAE is seeking clarification.
Source: ECEAE The European Coalition to End Animal Experiments
The next video explains why LUSH is so worried about REACH animal testing and why consumers should be aware of this largely unknown legislation!!!
Act now to save millions of animals from painful poisoning tests
In 2007, the European Union introduced the "REACH" regulation to provide for the Registration, Evaluation and Authorisation of Chemicals — more than 30,000 of them.
REACH requires a long list of animal tests, which means horrific suffering from chemical poisoning and death for millions of rabbits, mice and other creatures. But REACH also requires the use of animal testing alternatives where available, including measures to revise the regulation itself to reflect scientific progress on alternatives.
In April 2012, HSI submitted a comprehensive proposal to the European Commission calling for major changes to REACH testing requirements to spare millions of animals while providing the same level of regulatory scrutiny of chemicals.
We’re still waiting… and every day the Commission delays, more animals suffer and die needlessly.
Please sign HSI's petition to the European Commission today and join HSI in calling for immediate action to revise REACH to make full use of animal testing alternatives. HSI will hand in your signatures to Commissioners Potocnik and Tijani. Please click on the big blue button below to get redirected - the petition will open in a new tab.
REACH requires a long list of animal tests, which means horrific suffering from chemical poisoning and death for millions of rabbits, mice and other creatures. But REACH also requires the use of animal testing alternatives where available, including measures to revise the regulation itself to reflect scientific progress on alternatives.
In April 2012, HSI submitted a comprehensive proposal to the European Commission calling for major changes to REACH testing requirements to spare millions of animals while providing the same level of regulatory scrutiny of chemicals.
We’re still waiting… and every day the Commission delays, more animals suffer and die needlessly.
Please sign HSI's petition to the European Commission today and join HSI in calling for immediate action to revise REACH to make full use of animal testing alternatives. HSI will hand in your signatures to Commissioners Potocnik and Tijani. Please click on the big blue button below to get redirected - the petition will open in a new tab.
Thank you!