BOTOX
Botulinum toxin
Botulinum toxin is a protein produced by the bacterium Clostridium botulinum, and is considered the most powerful neurotoxin ever discovered. Botulinum toxin causes Botulism poisoning, a serious and life-threatening illness in humans and animals.
When introduced intravenously in monkeys, type A (Botox Cosmetic) of the toxin exhibits an LD50 of 40-56 ng, type C1 around 32 ng, type D 3200 ng, and type E 88 ng, rendering the above types some of the most powerful neurotoxins known.
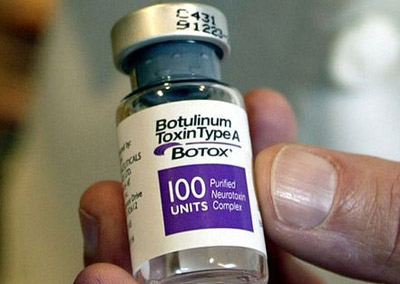
Popularly known by one of its trade names, Botox or Dysport or Xeomin, it is used for various cosmetic and medical procedures.
When introduced intravenously in monkeys, type A (Botox Cosmetic) of the toxin exhibits an LD50 of 40-56 ng, type C1 around 32 ng, type D 3200 ng, and type E 88 ng, rendering the above types some of the most powerful neurotoxins known.
Popularly known by one of its trade names, Botox or Dysport or Xeomin, it is used for various cosmetic and medical procedures.
History
Justinus Kerner described botulinum toxin as a "sausage poison" and "fatty poison", because the bacterium that produces the toxin often caused poisoning by growing in improperly handled or prepared meat products. It was Kerner, a physician, who first conceived a possible therapeutic use of botulinum toxin and coined the name botulism (from Latin botulus meaning "sausage"). In 1897, Emile van Ermengem identified the bacterium Clostridium botulinum to be the producer of botulinum toxin. In 1928, P. Tessmer Snipe and Hermann Sommer for the first time purified the toxin. In 1949, Arnold Burgen's group discovered, through an elegant experiment, that botulinum toxin blocks neuromuscular transmission through decreased acetylcholine release.
Therapeutic research
In the late 1960s Alan Scott, M.D., a San Francisco ophthalmologist, and Edward Schantz were the first to work on a standardized botulinum toxin preparation for therapeutic purposes. By 1973, Scott (now at Smith-Kettlewell Institute) used botulinum toxin type A (BTX-A) in monkey experiments, and, in 1980, he officially used BTX-A for the first time in humans to treat strabismus "crossed eyes", a condition in which the eyes are not properly aligned with each other, and "uncontrollable blinking" (blepharospasm). In 1993, Pasricha and colleagues showed that botulinum toxin could be used for the treatment of achalasia, a spasm of the lower esophageal sphincter. In 1994 Bushara showed that botulinum toxin injections inhibit sweating. This was the first demonstration of non-muscular use of BTX-A in humans.
Blepharospasm and strabismus
In the early 1980s, university-based ophthalmologists in the U.S.A. and Canada further refined the use of botulinum toxin as a therapeutic agent. By 1985, a scientific protocol of injection sites and dosage had been empirically determined for treatment of blepharospasm and strabismus.Side effects were deemed to be rare, mild and treatable. The beneficial effects of the injection lasted only 4–6 months. Thus, blepharospasm patients required re-injection two or three times a year.
In 1986, Scott's micro-manufacturer and distributor of Botox was no longer able to supply the drug because of an inability to obtain product liability insurance. Patients became desperate as supplies of Botox were gradually consumed, forcing him to abandon patients who would have been due for their next injection. For a period of four months, American blepharospasm patients had to arrange to have their injections performed by participating doctors at Canadian eye centers until the liability issues could be resolved.
In December 1989, Botox, manufactured by Allergan, Inc., was approved by the U.S. Food and Drug Administration (FDA) for the treatment of strabismus, blepharospasm, and hemifacial spasm in patients over 12 years old.
Cosmetic
The cosmetic effect of BTX-A on wrinkles was originally documented by a plastic surgeon from Sacramento, California, Dr. Richard Clark, and published in the journal Plastic and Reconstructive Surgery in 1989. Canadian husband and wife ophthalmologist and dermatologist physicians Carruthers JD and Carruthers JA were the first to publish a study on BTX-A for the treatment of glabellar frown lines in 1992. Similar effects had reportedly been observed by a number of independent groups (Brin, and the Columbia University group). After formal trials, on April 12, 2002, the FDA announced regulatory approval of botulinum toxin type A (Botox Cosmetic) to temporarily improve the appearance of moderate-to-severe frown lines between the eyebrows (glabellar lines). Subsequently, cosmetic use of botulinum toxin type A has become widespread with many celebrities viewing it as less intrusive and/or artificial than other types of plastic surgery. The results of cosmetic procedures vary but can last up to eight months. The U.S. Food and Drug Administration approved an alternative product-safety testing method in response to increasing public concern that LD50 testing was required for each batch sold in the market.
Source: Wikipedia
Justinus Kerner described botulinum toxin as a "sausage poison" and "fatty poison", because the bacterium that produces the toxin often caused poisoning by growing in improperly handled or prepared meat products. It was Kerner, a physician, who first conceived a possible therapeutic use of botulinum toxin and coined the name botulism (from Latin botulus meaning "sausage"). In 1897, Emile van Ermengem identified the bacterium Clostridium botulinum to be the producer of botulinum toxin. In 1928, P. Tessmer Snipe and Hermann Sommer for the first time purified the toxin. In 1949, Arnold Burgen's group discovered, through an elegant experiment, that botulinum toxin blocks neuromuscular transmission through decreased acetylcholine release.
Therapeutic research
In the late 1960s Alan Scott, M.D., a San Francisco ophthalmologist, and Edward Schantz were the first to work on a standardized botulinum toxin preparation for therapeutic purposes. By 1973, Scott (now at Smith-Kettlewell Institute) used botulinum toxin type A (BTX-A) in monkey experiments, and, in 1980, he officially used BTX-A for the first time in humans to treat strabismus "crossed eyes", a condition in which the eyes are not properly aligned with each other, and "uncontrollable blinking" (blepharospasm). In 1993, Pasricha and colleagues showed that botulinum toxin could be used for the treatment of achalasia, a spasm of the lower esophageal sphincter. In 1994 Bushara showed that botulinum toxin injections inhibit sweating. This was the first demonstration of non-muscular use of BTX-A in humans.
Blepharospasm and strabismus
In the early 1980s, university-based ophthalmologists in the U.S.A. and Canada further refined the use of botulinum toxin as a therapeutic agent. By 1985, a scientific protocol of injection sites and dosage had been empirically determined for treatment of blepharospasm and strabismus.Side effects were deemed to be rare, mild and treatable. The beneficial effects of the injection lasted only 4–6 months. Thus, blepharospasm patients required re-injection two or three times a year.
In 1986, Scott's micro-manufacturer and distributor of Botox was no longer able to supply the drug because of an inability to obtain product liability insurance. Patients became desperate as supplies of Botox were gradually consumed, forcing him to abandon patients who would have been due for their next injection. For a period of four months, American blepharospasm patients had to arrange to have their injections performed by participating doctors at Canadian eye centers until the liability issues could be resolved.
In December 1989, Botox, manufactured by Allergan, Inc., was approved by the U.S. Food and Drug Administration (FDA) for the treatment of strabismus, blepharospasm, and hemifacial spasm in patients over 12 years old.
Cosmetic
The cosmetic effect of BTX-A on wrinkles was originally documented by a plastic surgeon from Sacramento, California, Dr. Richard Clark, and published in the journal Plastic and Reconstructive Surgery in 1989. Canadian husband and wife ophthalmologist and dermatologist physicians Carruthers JD and Carruthers JA were the first to publish a study on BTX-A for the treatment of glabellar frown lines in 1992. Similar effects had reportedly been observed by a number of independent groups (Brin, and the Columbia University group). After formal trials, on April 12, 2002, the FDA announced regulatory approval of botulinum toxin type A (Botox Cosmetic) to temporarily improve the appearance of moderate-to-severe frown lines between the eyebrows (glabellar lines). Subsequently, cosmetic use of botulinum toxin type A has become widespread with many celebrities viewing it as less intrusive and/or artificial than other types of plastic surgery. The results of cosmetic procedures vary but can last up to eight months. The U.S. Food and Drug Administration approved an alternative product-safety testing method in response to increasing public concern that LD50 testing was required for each batch sold in the market.
Source: Wikipedia
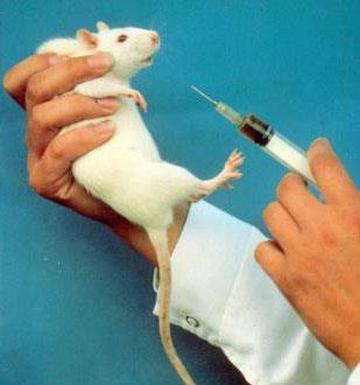
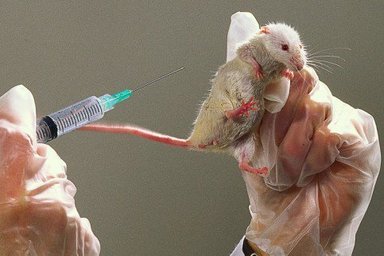
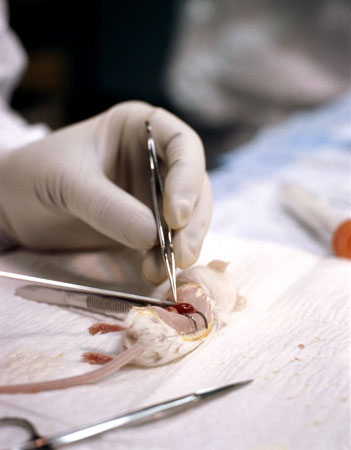
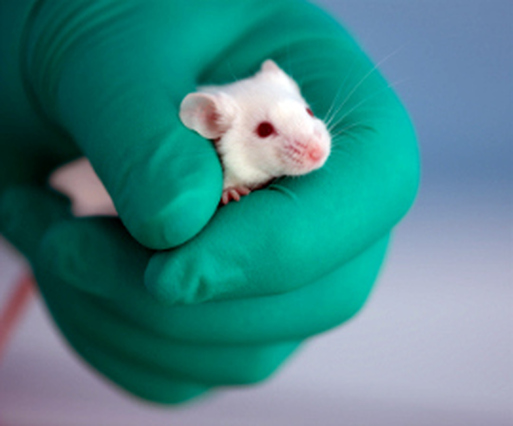
Thousands of mice are being poisoned to death to test the latest cosmetic craze: 'Botox'. In barbaric experiments known as LD50 toxicity tests - supposedly outlawed by the government in 1999 - the animals are injected with the toxin and suffer symptoms including impaired vision, paralysis of the body, and paralysis of the diaphragm, which leads to death by suffocation.
Following a rapid decrease in the overall number of LD50 tests in recent years, numbers are reported to have risen significantly over the last two years in line with the surge in popularity of botox injections. Apart from the devastating impact on the animal victims, the LD50 test is also a betrayal of people who imagine that botox cosmetic treatments have undergone proper safety tests. This is not the case.
The LD50 is now widely recognised in scientific and industry circles as being crude and unreliable. Given the highly toxic nature of botulinum, it is especially important that the substance is assessed in a scientific and dependable manner.
A battery of non-animal tests are already in existence which could, and should, immediately replace the animal-poisoning experiments.
Celebrity endorsement
Botox is the name given to botulinum toxin when used for cosmetic purposes. By paralysing the muscles, it makes it impossible to frown or crease the skin - hence making wrinkles 'disappear'. Botox injections are becoming increasingly popular as a cosmetic procedure, being popularised in no small measure thanks to celebrity endorsement. As well as its 'wrinkle-busting powers', botox has - apparently - been used by celebrities to paralyse the sweat glands under their arms, and thereby stop perspiration!
Biological weapon potential
Botulinum is considered to be more toxic to humans than most other known substances (it has biological weapon potential!). Its primary use is medical, in the treatment of various human conditions, including squints, involuntary twitches and facial spasms, with another 50 therapeutic applications currently being investigated. Cosmetic use, however, is the area that is growing the most rapidly. In the USA, it has been approved for medical reasons since 1989 and for cosmetic purposes since 2002. In the UK, it is also administered for both purposes, although it is not yet licensed for cosmetic use. The cosmetic application of botox, therefore, is categorised as 'off label', meaning that it is used at the discretion of the physician carrying out the procedure - and it is he or she who must take full personal responsibility for its safety, efficacy and quality. A UK-based company, called Dysport, has applied to the US Food and Drug Administration for a licence to market its own cosmetic brand of botox. If the licence is granted, the company will be able to advertise Dysport as a cosmetic product. This will inevitably lead to an even more significant increase in use.
Blanket licences
The potency of botulinum will vary from batch to batch. Before going on sale - whether for cosmetic or medical purposes - each batch will have gone through safety testing to assess its strength and toxicity. The Home Office supposedly no longer issues licences for testing cosmetic products on animals, as it does for pharmaceutical products. But because blanket licences are granted for botox toxicity testing, and no distinction is made between batches destined for the cosmetic as opposed to the pharmaceutical sector, it is all produced, and 'safety-tested' in the same way.
After potency testing on each batch by the manufacturers, a further confirmatory potency test is performed at one or more laboratories in the countries in which the batch will be used.
Mice suffocated to death
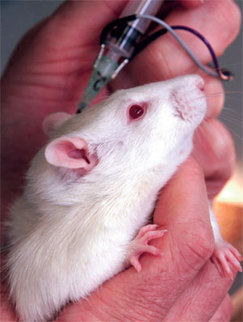
The potency test currently in use is the LD50 poisoning test. Acknowledged by the UK government to be a 'severe procedure', groups of mice are injected with various dilutions of toxin from a single batch to determine the dose that will kill half the mice in that group.
For each batch test, at least 100 mice are poisoned and observed for a period of 3-4 days. During this time, they will experience differing levels of muscular paralysis and impaired vision. Those injected with the highest dose will suffer paralysis of the diaphragm, leaving them unable to breathe and, consequently, they suffocate to death.
Accurate and humane non-animal test methods
Not only is the LD50 test appallingly cruel because of the numbers of animals used and the extreme suffering they undergo, it has long been acknowledged to be so crude as to be meaningless in terms of risk assessment for humans. The government announced in 1999 (after a legal challenge by the British Union for the Abolition of Vivisection) that LD50 tests would no longer be licensed except on 'exceptional scientific grounds'. This is because validated non-animal (in vitro) tests exist.
However, batch-testing of biological medicines - such as botulinum toxin - is one area in which the use of the LD50 is still permitted, despite the existence - in the case of botox - of a number of non-animal potency testing methods. These are not only humane, unlike LD50, they also give accurate and reproducible results.
Cosmetic botox boosts LD50 numbers
Claims that botulinum toxin is tested on animals primarily because of its therapeutic use ignore the fact that the biggest growth area is as an anti-wrinkle treatment.
In the years leading up to the partial ban on LD50 tests in 1999, the number of LD50 tests had been decreasing dramatically, and this trend should have continued. However, according to the Fund for the Replacement of Animals in Medical Experiments (FRAME), LD50 numbers increased significantly in 2001, with a further dramatic rise in 2002.
The rise correlates with a surge in the popularity of cosmetic botox. In the US, between 1998 and 2002, the use of Botox for cosmetic procedures increased by 1500%. According to the International Society of Aesthetic Plastic Surgeons, botox injections were the second most popular cosmetic procedure amongst women in 2001.
Government's double betrayal
So here we have the British government claiming that it has essentially 'banned' both LD50 tests AND cosmetics testing on animals, while continuing to licence (in breach of its own policy) LD50 testing of a cosmetic product! The licensing anomaly relating to botox means that large numbers of animals are paying the price of human vanity with severe suffering and, ultimately, their lives.
Animal Aid is calling upon the Home Office to insist that the industry implement non-animal test methods without delay - and to close the loophole permitting the testing on animals of a cosmetic product. Until that time, Animal Aid calls for a ban on the cosmetic use of botox. We further urge people considering botox injections to reflect on the fact that this dangerous toxin has not been adequately assessed for safety AND that many animals have been pointlessly abused and killed, in the manufacture of the product.
Source: Animal Aid UK
Corporate greed causes unnecessary cruelty to animals
by Dr Dan Lyons
September 7, 2012
Allergan, a Botox producer in Ireland, have developed a non-animal alternative to the horrific and unreliable LD50 test on mice that is applied to each batch of Botox. Great news?!
For the LD50 test, groups of mice are injected with different doses of Botulinum toxin into the abdomen. Many of the mice suffer from paralysis, impaired vision and respiratory distress. After up to three or four days of suffering, if left, they die of suffocation. Undercover investigations by the BUAV reveal that, in practice, attempts by staff to intervene and kill the mice before they died this agonising death often fail, with many more mice dying from the effects of the toxin instead of being killed before that happens.
I’ve been providing policy advice to the Irish Anti-Vivisection Society, and we’ve discovered that, sadly, Allergan have not yet shared their humane testing technology, so other companies like Ipsen and Merz are still doing the LD50 test, as reported by the Irish Examiner on 5 September 2012. It seems like Allergan would rather force their competitors to spend money developing their own non-animal test, rather than share their technology.
Legislation says that animal tests must be replaced by non-animal alternatives where ‘reasonably and practically available’. In practice, this non-animal alternative is available in these terms to Ipsen and Merz, they just need to take action with Allergan’s cooperation. The public are given the impression that the animal testing industry and government bust a gut to save animals from unnecessary testing and that if alternatives are available they will be used instead of animals. This episode will shock the public when they see how commercial greed is allowed to take precedence and obstruct the sharing of lifesaving technologies (which also happen to be much better tests in terms of improving the safety of these products for humans), especially when a large proportion of these unnecessary animal tests are for cosmetic Botulinum products.
The Irish Government have been very complacent on this – though that seems to be standard practice for national governments generally. It is their legal and moral duty to bring Allergan and the other companies together to spare thousands of animals from unnecessary abuse.
Source
Allergan, a Botox producer in Ireland, have developed a non-animal alternative to the horrific and unreliable LD50 test on mice that is applied to each batch of Botox. Great news?!
For the LD50 test, groups of mice are injected with different doses of Botulinum toxin into the abdomen. Many of the mice suffer from paralysis, impaired vision and respiratory distress. After up to three or four days of suffering, if left, they die of suffocation. Undercover investigations by the BUAV reveal that, in practice, attempts by staff to intervene and kill the mice before they died this agonising death often fail, with many more mice dying from the effects of the toxin instead of being killed before that happens.
I’ve been providing policy advice to the Irish Anti-Vivisection Society, and we’ve discovered that, sadly, Allergan have not yet shared their humane testing technology, so other companies like Ipsen and Merz are still doing the LD50 test, as reported by the Irish Examiner on 5 September 2012. It seems like Allergan would rather force their competitors to spend money developing their own non-animal test, rather than share their technology.
Legislation says that animal tests must be replaced by non-animal alternatives where ‘reasonably and practically available’. In practice, this non-animal alternative is available in these terms to Ipsen and Merz, they just need to take action with Allergan’s cooperation. The public are given the impression that the animal testing industry and government bust a gut to save animals from unnecessary testing and that if alternatives are available they will be used instead of animals. This episode will shock the public when they see how commercial greed is allowed to take precedence and obstruct the sharing of lifesaving technologies (which also happen to be much better tests in terms of improving the safety of these products for humans), especially when a large proportion of these unnecessary animal tests are for cosmetic Botulinum products.
The Irish Government have been very complacent on this – though that seems to be standard practice for national governments generally. It is their legal and moral duty to bring Allergan and the other companies together to spare thousands of animals from unnecessary abuse.
Source